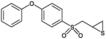
Competitive, mechanism-based inhibitor of matrix metalloproteinases 2 and 9 (MMP-2 and MMP-9). It possesses Ki values in the nanomolar range against the gelatinases (14nM for MMP-2; 600nM for MMP-9), but in the micromolar range against other metalloproteinases (206µM for MMP-1, 15µM for MMP-3, 96µM for MMP-7, and 4µM for ADAM17/TACE). This inhibitor has also been used in vitro, in vivo, and in tissue culture. In vivo, it is metabolized to an even more potent gelatinase inhibitor.
Product Details
| Formula: | C15H14O3S2 |
| |
| MW: | 306.4 |
| |
| CAS: | 292605-14-2 |
| |
| Purity: | ≥98% (TLC) |
| |
| Appearance: | White solid. |
| |
| Solubility: | Soluble in DMSO (at least 30mM) or 100% ethanol. |
| |
| Shipping: | Blue Ice |
| |
| Long Term Storage: | -20°C |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
ADAM17 Mediates Proteolytic Maturation of Voltage-Gated Calcium Channel Auxiliary α2δ Subunits, and Enables Calcium Current Enhancement: I. Kadurin, et al.; Function (Oxf.)
3, zqac013 (2022),
Abstract;
Primitive macrophages control HSPC mobilization and definitive haematopoiesis: J. Travnickova, et al.; Nat. Commun.
6, 6227 (2015),
Application(s): Cell Culture, Assay,
Abstract;
Metabolism of a highly selective gelatinase inhibitor generates active metabolite: M. Lee et al.; Chem. Biol. Drug. Des.
70, 371 (2007),
Abstract;
Inhibition of human prostate cancer growth, osteolysis and angiogenesis in a bone metastasis model by a novel mechanism-based selective gelatinase inhibitor: R. D. Bonfil et al.; Cancer Res.
118, 2721 (2006),
Abstract;
Antimetastatic activity of a novel mechanism-based gelatinase inhibitor: A. Krüger, et al.; Cancer Res.
65, 3523 (2005),
Abstract;
Full Text
A Convenient Synthesis of a Selective Gelatinase Inhibitor as an Antimetastatic Agent: I.T. Lim, et al.; J. Org. Chem.
69, 3572 (2004),
Abstract;
Pronounced diversity in electronic and chemical properties between the catalytic zinc sites of tumor necrosis factor-alpha-converting enzyme and matrix metalloproteinases despite their high structural similarity: A. Solomon et al.; J. Biol. Chem.
279, 31646 (2004),
Abstract;
Complex pattern of membrane type 1 matrix metalloproteinase shedding. Regulation by autocatalytic cells surface inactivation of active enzyme: M. Toth et al.; J. Biol. Chem.
277, 26340 (2002),
Abstract;
X-ray absorption studies of human matrix metalloproteinase-2 (MMP-2) bound to a highly selective mechanism-based inhibitor. comparison with the latent and active forms of the enzyme: O. Kleifeld et al.; J. Biol. Chem.
276, 17125 (2001),
Abstract;
Potent and Selective Mechanism-Based Inhibition of Gelatinases: S. Brown et al.; J. Am. Chem. Soc.
122, 6799 (2000),
Abstract;