Replaces Prod. #: ALX-270-243
High affinity cannabinoid receptor (CB) agonist (Ki=62.3nM and 3.3nM at the human cloned CB1 and CB2 receptors, respectively). Potent analgesic in a rat model of neuropathic pain. Activates p42 and p44 MAP kinase via a receptor-mediated signaling.
Product Details
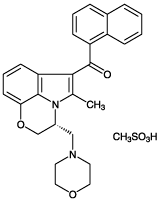
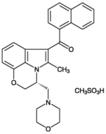
| Alternative Name: | (R)-(+)-[2,3-Dihydro-5-methyl-3-(4-morpholinylmethyl)pyrrolo[1,2,3-de]-1,4-benzoxazin-6-yl]-1-naphthalenylmethanone . mesylate |
| |
| Formula: | C27H26N2O3 . CH3SO3H |
| |
| MW: | 522.6 |
| |
| CAS: | 131543-23-2 |
| |
| Purity: | ≥98% (HPLC) |
| |
| Appearance: | White to off-white solid. |
| |
| Solubility: | Soluble in DMSO; insoluble in water. |
| |
| Shipping: | Ambient Temperature |
| |
| Long Term Storage: | -20°C |
| |
| Use/Stability: | Stable for at least 1 year after receipt when stored, as supplied, at -20°C. Stock solutions are stable for up to 3 months at -20°C. |
| |
| Technical Info/Product Notes: | WIN-55,212-2 is a controlled substance in some countries and may require an import/export license from the local authorities depending on the country of import. Orders for this product will incur an administrative fee to cover license expenses. Please inquire for additional information. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Cannabinoid functions in the amygdala contribute to conditioned fear memory in streptozotocin-induced diabetic mice: Interaction with glutamatergic functions: H. Ikeda, et al.; Exp. Neurol.
269, 233 (2015),
Application(s): Injection into mice,
Abstract;
Immunoregulation of a viral model of multiple sclerosis using the synthetic cannabinoid R+WIN55,212: J.L. Croxford & S.D. Miller ; J. Clin. Invest.
111, 1231 (2003),
Abstract;
Anti-tumoral action of cannabinoids: involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation: I. Galve-Roperh, et al.; Nat. Med.
6, 313 (2000),
Abstract;
An analgesia circuit activated by cannabinoids: I.D. Meng, et al.; Nature
395, 6700 (1998),
Abstract;
The analgesic effects of R(+)-WIN 55,212-2 mesylate, a high affinity cannabinoid agonist, in a rat model of neuropathic pain: U. Herzberg, et al.; Neurosci. Lett.
221, 157 (1997),
Abstract;
Rat brain cannabinoid receptor modulates N-type Ca2+ channels in a neuronal expression system: X. Pan, et al.; Mol. Pharmacol.
49, 707 (1996),
Abstract;
Activation of mitogen-activated protein kinases by stimulation of the central cannabinoid receptor CB1: M. Bouaboula, et al.; Biochem. J.
312, 637 (1995),
Abstract;
Comparison of the pharmacology and signal transduction of the human cannabinoid CB1 and CB2 receptors: C.C. Felder, et al.; Mol. Pharmacol.
48, 443 (1995),
Abstract;
Inhibition of long-term potentiation in rat hippocampal slices by anandamide and WIN55212-2: reversal by SR141716 A, a selective antagonist of CB1 cannabinoid receptors: J.P. Terranova, et al.; Naunyn-Schmiedeberg's Arch. Pharmacol.
352, 576 (1995),
Abstract;
(+)-WIN 55,212-2, a novel cannabinoid receptor agonist, exerts antidystonic effects in mutant dystonic hamsters: A. Richter & W. Loscher; Eur. J. Pharmacol.
264, 371 (1994),
Abstract;
Conformationally restrained analogues of pravadoline: nanomolar potent, enantioselective, (aminoalkyl)indole agonists of the cannabinoid receptor: T.E. D'Ambra, et al.; J. Med. Chem.
35, 124 (1992),
Abstract;