Product Details
| Alternative Name: | Microtubule-associated protein 1A/1B-light chain 3 |
| |
| MW: | ~16kDa (predicted), 20kDa (observed) |
| |
| Source: | Produced in E. coli. Human LC3-I is fused at the N-terminus to a His-tag. |
| |
| UniProt ID: | Q9H492 (LC3A), Q9GZQ8 (LC3B), Q9BXW4 (LC3C) |
| |
| Formulation: | Liquid. In 25mM TRIS, pH 7.5, containing 250mM sodium chloride, 1.0mM EDTA, and 1.0mM DTT. |
| |
| Purity: | ≥95% (SDS-PAGE) |
| |
| Purity Detail: | Purified by multi-step chromatography. |
| |
| Endotoxin Content: | <50EU/mg purified protein (LAL test) |
| |
| Applications: | WB
|
| |
| Application Notes: | Western blot control. |
| |
| Shipping: | Dry Ice |
| |
| Long Term Storage: | -80°C |
| |
| Scientific Background: | The generic term ‘‘autophagy’’ comprises several processes by which the lysosome acquires cytosolic cargo, with three types of autophagy being discerned in the literature: (1) macroautophagy, characterized by the formation of a crescent-shaped structure (the phagophore) that expands to form the double-membrane autophagosome, capable of fusion with the lysosome; (2) microautophagy, in which lysosomes invaginate and directly sequester cytosolic components; and (3) chaperone-mediated autophagy (CMA), which involves translocation of unfolded proteins across the lysosomal membrane. Upregulation of autophagy pathways occurs in response to extra- or intracellular stress and signals such as starvation, growth factor deprivation, ER stress and pathogen infection. Malfunction of these pathways is linked to various human pathologies including cancer, neurodegeneration and infectious diseases. Selective macroautophagy describes the pathway of self-degradation of whole cellular components, protein aggregates or unusually long-lived proteins; in which double-membrane autophagosomes sequester organelles, ubiquitinylated proteins or ubiquitinylated protein aggregates and subsequently fuse with lysosomes for breakdown by resident hydrolases. Formation and expansion of the pre-autophagosomal structure requires the attachment of the ubiquitin-like protein ATG8 via its C-terminal glycine to the amino group of phosphatidylethanolamine (PE), enabling its anchoring to the isolation membrane of the autophagosome. In mammals, ATG8 is represented by at least seven related proteins that fall into two subgroups, LC3- and GABARAP-like proteins. The free and PE linked versions of these proteins are often referred to as LC3-I and LC3-II respectively. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
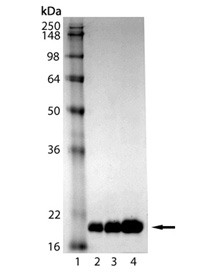
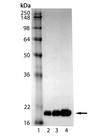
SDS-PAGE analysis of LC3-I (human), (recombinant) (Prod. no. ADI-APR-100): Lane 1: MW Marker, Lane 2: 1 µg, Lane 3: 2 µg, Lane 4: 5 µg LC3-I.
Please mouse over
Product Literature References
Atg5 Disassociates the V1V0-ATPase to Promote Exosome Production and Tumor Metastasis Independent of Canonical Macroautophagy: H. Guo, et al.; Dev. Cell
43, 716 (2017),
Abstract;
Hepatitis B virus-triggered autophagy targets TNFRSF10B/death receptor 5 for degradation to limit TNFSF10/TRAIL response: G.C. Shin, et al.; Autophagy
12, 2451 (2016),
Abstract;
Full Text
Autophagic flux and oxidative capacity of skeletal muscles during acute starvation: M. Mofarrahi, et al.; Autophagy
9, 1604 (2013),
Abstract;
General Literature References
Selective autophagy mediated by autophagic adapter proteins: T. Johansen & T. Lamark; Autophagy
7, 279 (2011),
Abstract;
A role for ubiquitin in selective autophagy: V. Kirkin, et al.; Mol. Cell
34, 259 (2009),
Abstract;
Regulation mechanisms and signaling pathways of autophagy: C. He & D.J. Klionsky; Annu. Rev. Genet.
43, 67 (2009),
Abstract;
Autophagy fights disease through cellular self-digestion: N. Mizushima, et al.; Nature
451, 1069 (2008),
Abstract;
The Atg8 and Atg12 ubiquitin-like conjugation systems in macroautophagy. 'Protein modifications: beyond the usual suspects' review series: J. Geng & D.J. Klionsky; EMBO Rep.
9, 859 (2008),
Abstract;