A subunit of the 26S proteasome, S5a, recognises and binds multi-ubiquitinylated proteins containing chains of at least four ubiquitin moieties. Purified recombinant S5a retains the ability to bind multi-ubiquitinylated proteins in isolation, when immobilised on glutathione-Sepharose, and after being eluted from glutathione-Sepharose, run on SDS-PAGE, and blotted onto nitrocellulose. Human S5a sequence, with an N-terminal fusion of glutathione S-transferase (Schistosoma japonicum), was expressed in E. coli with GST tag at amino terminus.
Product Details
| Alternative Name: | 26S proteasome non-ATPase regulatory subunit 4, Antisecretory factor 1, Multiubiquitin chain-binding protein |
| |
| MW: | ~78kDa (observed) |
| |
| Source: | Produced in E. coli. |
| |
| UniProt ID: | P55036 |
| |
| Formulation: | Liquid. In 20mM TRIS, pH 7.5, containing 500mM sodium chloride. |
| |
| Purity: | ≥95% (SDS-PAGE) |
| |
| Purity Detail: | Purified by multi-step chromatography. |
| |
| Appearance: | Colorless liquid. |
| |
| Activity: | Bound to mutliubiquitin chains of length >4 when conjugated to agarose (Prod. No. BML-UW8635). |
| |
| Application Notes: | Useful for the isolation of multi-ubiquitinylated proteins. |
| |
| Shipping: | Dry Ice |
| |
| Long Term Storage: | -80°C |
| |
| Scientific Background: | S5a (RPN10), a subunit of the 19S regulator of the 26S proteasome, binds multi-ubiquitinylated proteins containing chains of at least four ubiquitin moieties. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |

Western Blot Analysis of Rpn10/S5a (Prod. No. BML-UW8465). Lane 1: MW Marker, Lane 2: Rpn10/S5a, Lane 3: Multiubiquitin chains (Prod. No. BML-UW8860), Lane 4: HeLa lysate (Prod. No. ADI-LYC-HL100). The blot on the left was probed with 10 µg/ml Rpn10, right with blocking buffer only. Rpn10 was detected by probing with GST polyclonal antibodies.
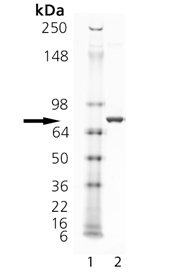
SDS-PAGE Analysis of Rpn10/S5a. Lane 1: MW Marker, Lane 2: 1 µg.
Please mouse over
Product Literature References
Quantitative Affinity Interaction of Ubiquitinated and Non-ubiquitinated Proteins with Proteasome Subunit Rpn10: O. A. Buneeva, et al.; Biochemistry (Moscow)
82, 1042 (2017),
Abstract;
Full Text