| Alternative Name: | cAMP-dependent protein kinase catalytic subunit β, Protein kinase A C-β |
| |
| MW: | ~65kDa |
| |
| Source: | Produced in Sf9 insect cells using an N-terminal GST-tag. Produced in a baculovirus expression system. |
| |
| UniProt ID: | P22694 |
| |
| Formulation: | Liquid. In 50mM Tris-HCl, pH 7.5, containing 150mM sodium chloride, 0.25mM DTT, 0.1mM EGTA, 0.1mM EDTA, 0.1mM PMSF, and 25% glycerol. |
| |
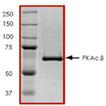
| Purity: | ≥80% (SDS-PAGE) |
| |
| Purity Detail: | Purified by SDS-PAGE / densitometry. |
| |
| Applications: | Kinase assay
|
| |
| Shipping: | Dry Ice |
| |
| Long Term Storage: | -80°C |
| |
| Scientific Background: | PKA belongs to the cAMP-dependent protein kinase (PKA) family. PKA isoforms type I and II include respective dimeric R subunits RI and RII which each produce two subunits designated RI-α, RI-β, RII-α, and RII-β. The catalytic subunit also consists of more than one gene product. Researchers cloned mammalian catalytic subunit C-α, C-β, and C-γ cDNAs. The catalytic subunit C-β belongs to the Ser/Thr protein kinase family. Activated by cAMP, PKA C-β resides in the cytoplasm (inactive holoenzyme and monomeric catalytic subunit), and translocates into the nucleus (monomeric catalytic subunit). A number of inactive tetrameric holoenzymes result from the combination of homo- or heterodimers of the different regulatory subunits associated with the two catalytic subunits. cAMP causes the dissociation of the inactive holoenzyme into a dimer of regulatory subunits bound to four cAMP and two free monomeric catalytic subunits. PKA C-β acts as a p75 neurotrophin receptor (NTR)-interacting protein, which phosphorylates p75 (NTR) at Ser304. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |