Product Details
| Alternative Name: | GST-π, GST-P, Glutathione S-transferase P, GSTP1 |
| |
| Clone: | USal-hGST-Pi-McAb-1 |
| |
| Host: | Mouse |
| |
| Isotype: | IgG1 |
| |
| Immunogen: | Recombinant human placenta GST-Pi (glutathione S-transferase-Pi). |
| |
| UniProt ID: | P09211 |
| |
| Species reactivity: | Human, Rat
Leishmania
|
| |
| Crossreactivity: | Does not cross-react with human or rat class alpha GST. |
| |
| Applications: | IHC (FS), WB
|
| |
| Recommended Dilutions/Conditions: | Immunohistochemistry (frozen sections, 1:100)
Western Blot (1:150-1:1,000)
Suggested dilutions/conditions may not be available for all applications.
Optimal conditions must be determined individually for each application. |
| |
| Positive Control: | Rat hepatocarcinoma can be used as positive control. |
| |
| Purity Detail: | Protein G-affinity purified. |
| |
| Formulation: | Liquid. Contains 100nM sodium azide and 0.7% BSA. |
| |
| Handling: | Avoid freeze/thaw cycles. After opening, prepare aliquots and store at -20°C. |
| |
| Shipping: | Blue Ice |
| |
| Short Term Storage: | +4°C |
| |
| Long Term Storage: | -20°C |
| |
| Scientific Background: | The GST Family
Glutathione S-transferases (GSTs) are a large multigen family of multifunctional enzymes, which play an important role in detoxification of potentially genotoxic chemicals, by catalyzing the conjugation of glutathione to a large number of hydrophobic and electrophilic compounds, including carcinogens. They also bind to a variety of nonsubstrate ligands. The GSTs are widely distributed in mammalian species and can be grouped into four classes, based on their biochemical, immunological and structural properties: alpha, mu, pi and theta.
GSTs are dimeric, mainly cytosolic, enzymes that have extensive ligand binding properties in addition to their catalytic role in detoxification. A separate microsomal class of GSTs exists which is quite distinct from the cytosolic GSTs, and is designated as "membrane-associated proteins in eicosanoid and glutathione metabolism" (MAPEG). The N-terminal region tends to be better conserved within classes as it includes an important part of the active site.GSTs are believed to play a role in cancer prevention, as the electrophilic compounds that are conjugated to glutathione could otherwise react with and modify cellular components such as DNA, thereby initiating carcinogenesis. On the other hand, GSTs might detoxify chemotherapeutic drugs in cancer cells, thus contributing to drug resistance.
In summary, GSTs are a group of enzymes which are centrally involved in drug metabolism and detoxification. Because of their role in detoxification, they have been implicated in drug sensitivity and resistance. They may also influence mutagenesis and carcinogenesis and thus, increased GST activity may become a useful cancer marker.
Since many GST genes are polymorphic, there has been considerable interest in determining whether particular allelic variants are also associated with an increase of risk for certain diseases.
Glutathione S-transferase-Pi
The GST-Pi class (phase II detoxification enzymes) is the most abundant of the human glutathione S-transferases family, a major group of detoxification enzymes and widely distributed in the human body.
In normal human tissues, the enzymes protect cells against reactions with glutathione and reactive oxygen species by reducing organic hydroperoxides via gluthathione peroxidase activity.
The role of GST-Pi in tumor growth and progression is less well known. It is reported to be variably expressed in breast cancer and is associated with estrogen receptor levels expressed by the tumor. Down-regulation of GST-Pi activity in a study done on a T cell line appears to favor apoptosis and inhibition of GST-Pi function induces apoptosis in rat hepatoma cells.
The importance in cancer research is clear, as GST-Pi has been implicated in protection against apoptosis and also suggested to have a role in jun kinase inhibition.
The majority of human tumor cell lines express significant amounts of class pi GST, which is why it is being investigated as a potential marker for various types of cancer. GST-Pi is thought to be associated with increased resistance to anticancer drugs. GST-Pi expression is under investigation as a prognostic indicator for resistance to chemotherapy and a marker of treatment resistance. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |

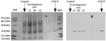
Figure 1: Left: Polyacrylamide gel electrophoresis (SDS-PAGE) and Coomassie brilliant blue staining of molecular weight standard (MW), pure GST-Pi protein and liver homogenate (5µg protein) from normal rat liver (Control) or from liver tissue containing neoplastic lesions at different time points during hepatocarcinogenesis. Right: Western blot using anti-GST-Pi MAb.

Figure 2: Immunohistochemistry of rat liver at 12, 20 or 32 weeks after initiation of liver carcinogenesis. GST-Pi-positive tissue was stained with an anti-GST-Pi MAb in a 30µm thick rat liver slice. Original magnification 4x.
Please mouse over
Product Literature References
Bile acid secretion during rat liver carcinogenesis: M.J. Monte, et al.; Life Sci.
66, 1085 (2000),
Abstract;