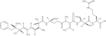Linear nodularin has been demonstrated to be a biosynthetic precursor of the highly toxic cyclic pentapeptide, nodularin. It is an intermediate of biodegradation by natural populations of bacteria in aquatic samples.
Product Details
| Formula: | C41H62N8O11 |
| |
| MW: | 843.0 |
| |
| Source: | Isolated from Nodularia spumigena. |
| |
| RTECS: | GU2294250 |
| |
| Purity: | ≥95% (HPLC) |
| |
| Identity: | Identity determined by MS. |
| |
| Appearance: | Pale powder or an oil (film), depending on the lot. |
| |
| Solubility: | Soluble in methanol:water (1:1). |
| |
| Shipping: | Ambient Temperature |
| |
| Long Term Storage: | -20°C |
| |
| Technical Info/Product Notes: | Not toxic to mice at 2 mg/kg I.P. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
An ELISA-based Method for Variant-independent Detection of Total 3 Microcystins and Nodularins via Multi-immunogen Approach: J. Liu, et al.; Environ. Sci. Technol.
55, 12984 (2021),
Abstract;
Biodegradation of microcystins and nodularin in freshwaters: C. Edwards, et al.; Chemosphere
73, 1315 (2008),
Abstract;
Characterization of nodularin variants in Nodularia spumigena from the Baltic Sea using liquid chromatography/mass spectrometry/mass spectrometry: H. Mazur-Marzec, et al.; Rapid Commun. Mass Spectrom.
20, 2023 (2006),
Abstract;
Nodularin-induced genotoxicity following oxidative DNA damage and aneuploidy in HepG2 cells: A. Lankoff, et al.; Toxicol. Lett.
164, 239 (2006),
Abstract;
Bacterial degradation of microcystins and nodularin: S. Imanishi, et al.; Chem. Res. Toxicol.
18, 591 (2005),
Abstract;
New Nodularins: A General Method for Structure Assignment: M. Namikoshi, et al.; J. Org. Chem.
59, 2349 (1994),
Full Text
Structure and Biosynthesis of toxins from blue-green algae: K.L. Rinehart, et al.; J. Appl. Phycol. 6, 159 (1994),
Isolation of linear peptides related to the hepatotoxins nodularin and microcystins: B.W. Choi, et al; Tetrahed. Lett. 34, 7881 (1993),
Related Products