Drug-induced acute kidney injury account for as much as 25% of all episodes of acute renal failure - a serious problem in clinical medicine as well as in the pharmaceutical industry. In drug development, some therapeutic candidates fail due to unexpected toxic effects in the kidneys, resulting in adverse outcomes and ultimately development termination of these candidates (attrition).
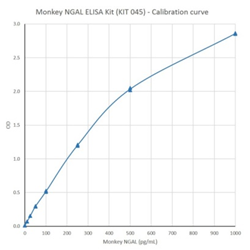
NGAL provides an excellent endogenous biomarker for acute kidney injury (AKI) which occurs prior to ARF. Changes in NGAL urine and blood can be used to assess kidney status as a supplement to other established medical products or procedures.
“An appropriate preclinical biomarker-based strategy facilitates selection of drug candidates with improved kidney safety profile” - Burt D et al. (2013) Drug Chem Toxicol. Oct 16. Epub ahead of print