Product Details
| Alternative Name: | HSP84, Heat shock protein 90β |
| |
| Host: | Rabbit |
| |
| Immunogen: | Recombinant human HSP90β. |
| |
| UniProt ID: | P08238 |
| |
| Source: | Purified from rabbit serum. |
| |
| Species reactivity: | Human, Mouse
Bovine, Dog, Guinea pig, Hamster, Porcine, Rabbit, Sheep
|
| |
| Applications: | Flow Cytometry
|
| |
| Recommended Dilutions/Conditions: | Flow Cytometry (1:100)
Suggested dilutions/conditions may not be available for all applications.
Optimal conditions must be determined individually for each application. |
| |
| Application Notes: | Detects a band of ~90kDa by Western blot. |
| |
| Purity Detail: | Protein A affinity purified. |
| |
| Formulation: | Liquid. In PBS containing 0.09% sodium azide. |
| |
| Handling: | Avoid freeze/thaw cycles. Protect from light. |
| |
| Shipping: | Blue Ice Not Frozen |
| |
| Long Term Storage: | +4°C |
| |
| Scientific Background: | The 90 kDa molecular chaperone family includes 90 kDa heat shock protein Hsp90 and 94 kDa glucose-regulated protein grp94, both major molecular chaperones of the cytosol and the endoplasmic reticulum. Mammalian cells express inducible Hsp90α and constitutive Hsp90β isoforms that are encoded by separate genes. The amino acid sequences of human and yeast Hsp90α are 85% and 90% homologous to those of Hsp90β, respectively. All known members of the Hsp90 protein family are highly conserved, especially in the N-terminal and C-terminal regions containing independent chaperone sites with different substrate specificity. These ubiquitous and highly conserved proteins account for 1-2% of all cellular protein in most cells. Hsp90 functions as part of the cell's powerful network of chaperones to fight the deleterious consequences of protein unfolding caused by non-physiological conditions. In the absence of stress, however, Hsp90 provides a necessary component of such fundamental cellular processes as hormone signaling and cell cycle control by serving as a chaperone for many key signaling molecules including steroid receptors, cell cycle kinases involved in signal transduction, and p53. As many of these client proteins are known oncogenes, Hsp90 inhibitors such as 17-AAG, a geldanamycin analog, have been of benefit in the treatment of many cancers. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
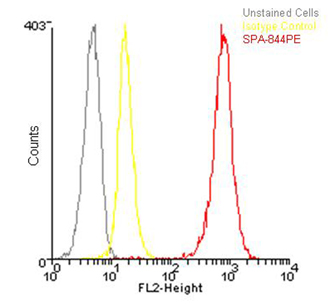
Flow cytometry analysis of 106 Jurkat cells using Hsp90β, pAb (PE conjugate) at a concentration of 50µg/ml.
Please mouse over
Product Literature References
Gas Flow Shaping via Novel Modular Nozzle System (MoNoS) Augments kINPen-Mediated Toxicity and Immunogenicity in Tumor Organoids: J. Berner, et al.; Cancers
15, 1254 (2023),
Abstract;
General Literature References
Drugging the cancer chaperone HSP90: combinatorial therapeutic exploitation of oncogene addiction and tumor stress: P. Workman, et al.; Ann. N.Y. Acad. Sci.
1113, 202 (2007),
Abstract;
The charged region of Hsp90 modulates the function of the N-terminal domain: T. Scheibel, et al.; PNAS USA
96, 1297 (1999),
Abstract;
Hsp90 as a capacitor for morphological evolution: S.L. Rutherford & S. Lindquist; Nature
396, 336 (1998),
Abstract;
The Hsp90 complex--a super-chaperone machine as a novel drug target: T. Scheibel & J. Buchner; Biochem. Pharmacol.
56, 675 (1998),
Abstract;
Guidebook to Chaperones: T. Scheibel & J. Buchner; Ed. Gething, M.J. Oxford Univ. Press 147 (1997), Book,
Related Products










