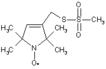
Highly reactive thiol-specific spin label. Has been used to label cysteine residues in proteins (site-directed labeling, SDS-labeling). Allows protein structure and protein dynamics determination as well as the study of protein-protein and protein-oligonucleotide interactions.
Product Details
| Alternative Name: | (1-Oxyl-2,2,5,5-tetramethylpyrroline-3-methyl) methanethiosulfonate |
| |
| Formula: | C10H18NO3S2 |
| |
| MW: | 264.3 |
| |
| CAS: | 81213-52-7 |
| |
| Purity: | ≥98% (HPLC) |
| |
| Appearance: | Yellow crystalline solid. |
| |
| Solubility: | Soluble in water, methanol, 100% ethanol, DMSO, acetonitrile or acetone. |
| |
| Shipping: | Blue Ice |
| |
| Long Term Storage: | -20°C |
| |
| Handling: | Protect from light. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Rapid Scan Electron Paramagnetic Resonance Spectroscopy Is a Suitable Tool to Study Intermolecular Interactions of Intrinsically Disordered Protein: J. Dröden, et al.; Biology
12, 79 (2023),
Abstract;
Location of the cross-β structure in prion fibrils: A search by seeding and electron spin resonance spectroscopy: B.K.Y. Chu, et al.; Protein Sci.
31, e4326 (2022),
Abstract;
Nanodisc Lipids Exhibit Singular Behaviors Implying Critical Phenomena: P.S. Ho, et al.; Langmuir
49, 15372 (2022),
Abstract;
Proton-driven alternating access in a spinster lipid transporter: R. Dastvan, et al.; Nat. Commun.
13, 5161 (2022),
Abstract;
Structure and mechanism of human cystine exporter cystinosin: X. Guo, et al.; Cell
185, 3739 (2022),
Abstract;
Protein and solutes freeze-concentration in water/glycerol mixtures revealed by pulse EPR: N. Isaev, et al.; Eur. J. Pharm. Biopharm.
169, 44 (2021),
Abstract;
Conformational transitions of the sodium-dependent sugar transporter, vSGLT: A. Paz, et al.; PNAS
115, E2742 (2018),
Abstract;
Full Text
A new structural model of Alzheimer's Aβ42 fibrils based on electron paramagnetic resonance data and Rosetta modeling: L. Gu, et al.; J. Struct. Biol.
194, 61 (2016),
Application(s): Cell culture,
Abstract;
Lipids modulate the conformational dynamics of a secondary multidrug transporter: C. Martens, et al.; Nat. Struct. Mol. Biol.
23, 744 (2016),
Application(s): Spin-labeling, after LmrP mutant purification,
Abstract;
Protonation-dependent conformational dynamics of the multidrug transporter EmrE: R. Dastvan, et al.; PNAS
113, 1220 (2016),
Abstract;
Full Text
Conformational dynamics of the nucleotide binding domains and the power stroke of a heterodimeric ABC transporter: S. Mishra et al.; eLife
3, e02740 (2014),
Application(s): Size-exclusion chromatography,
Abstract;
Full Text
Alzheimer's Aβ42 and Aβ40 peptides form interlaced amyloid fibrils: L. Gu, et al.; J. Neurochem.
126, 305 (2013),
Application(s): MALDI-TOF mass spectrometry,
Abstract;
Full Text
The Structure of the RLIP76 RhoGAP-Ral Binding Domain Dyad: Fixed Position of the Domains Leads to Dual Engagement of Small G Proteins at the Membrane: K.V. Rajasekar, et al.; Structure
21, 2131 (2013),
Application(s): Mass spectrometry, Assay,
Abstract;
Full Text
Hierarchical Organization in the Amyloid Core of Yeast Prion Protein Ure2: S. Ngo, et al.; J. Biol. Chem.
286, 29691 (2011),
Application(s): MALDI-TOF mass spectrometry,
Abstract;
Full Text
Calcium structural transition of human cardiac troponin C in reconstituted muscle fibres as studied by site-directed spin labelling: M. Nakamura, et al.; J. Mol. Biol.
348, 127 (2005),
Abstract;
Inter- and intra-molecular distances determined by EPR spectroscopy and site-directed spin labeling reveal protein-protein and protein-oligonucleotide interaction: H.J. Steinhoff; Biol. Chem.
385, 913 (2004),
Abstract;
Spontaneous refolding of the pore-forming Colicin A toxin upon membrane association as studied by X-band and W-band high-field electron paramagnetic resonance spectroscopy: A. Savitski, et al.; J. Phys. Chem. B 108, 9541 (2004),
Methods for study of protein dynamics and protein-protein interaction in protein-ubiquitination by electron paramagnetic resonance spectroscopy: H.J. Steinhoff; Front. Biosci.
7, c97 (2002),
Abstract;
Protein structure determination using long-distance constraints from double-quantum coherence ESR: study of T4 lysozyme: P.P. Borbat, et al.; JACS
124, 5304 (2002),
Abstract;
Pressure-induced thermostabilization of glutamate dehydrogenase from the hyperthermophile Pyrococcus furiosus: M.M. Sun, et al.; Protein Sci.
8, 1056 (1999),
Abstract;
A novel reversible thiol-specific spin label: papain active site labeling and inhibition: L.J. Berliner, et al.; Anal. Biochem.
119, 450 (1982),
Abstract;