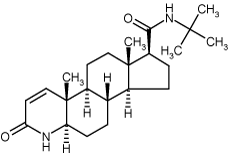
Inhibitor of 5α-reductase, the enzyme which converts testosterone to the more potent androgen, 5α-dihydrotestosterone. Used as a treatment in benign prostatic hyperplasia and prostate cancer. Also used to treat male pattern hair loss.
Product Details
| Alternative Name: | MK-906 |
| |
| Formula: | C23H36N2O2 |
| |
| MW: | 372.5 |
| |
| CAS: | 98319-26-7 |
| |
| MI: | 14: 4082 |
| |
| RTECS: | CL5245000 |
| |
| Purity: | ≥98% (Assay (UV)) |
| |
| Identity: | Determined by IR. |
| |
| Appearance: | White to off-white crystalline powder. |
| |
| Solubility: | Soluble in DMSO, 100% ethanol, methanol or propanol. |
| |
| Shipping: | Ambient Temperature |
| |
| Long Term Storage: | +4°C |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Effects of sex steroid hormones and their metabolites on neuronal injury caused by oxygen-glucose deprivation/reoxygenation in organotypic hippocampal slice cultures: Y. Ishihara, et al.; Steroids
113, 71 (2016),
Application(s): Examination of the protective mechanism of progesterone against OGD/reoxygenation-induced neurotoxicity,
Abstract;
Anti-nociceptive and anti-inflammatory properties of 5alpha-reductase inhibitor finasteride in experimental animals: D. Duborija-Kovacevic, et al.; Eur. J. Drug Metab. Pharmacokinet.
33, 181 (2008),
Abstract;
Finasteride for prostatic disease: an updated and comprehensive review: C. De Nunzio, et al.; Expert Opin. Drug Metab. Toxicol.
4, 1561 (2008), (Review),
Abstract;
Finasteride induces apoptosis via Bcl-2, Bcl-xL, Bax and caspase-3 proteins in LNCaP human prostate cancer cell line: J.M. Golbano, et al.; Int. J. Oncol.
32, 919 (2008),
Abstract;
A new look at the 5alpha-reductase inhibitor finasteride: D.A. Finn, et al.; CNS Drug Rev.
12, 53 (2006), (Review),
Abstract;
Finasteride in the treatment of alopecia: J.F. Libecco & W.F. Bergfeld; Expert Opin. Pharmacother.
5, 933 (2004), (Review),
Abstract;
Finasteride: the first 5 alpha-reductase inhibitor: S.L. Sudduth & M.J. Koronkowski; Pharmacotherapy
13, 309 (1993), (Review),
Abstract;
Azasteroids: structure-activity relationships for inhibition of 5 alpha-reductase and of androgen receptor binding: G.H. Rasmusson, et al.; J. Med. Chem.
29, 2298 (1986),
Abstract;