Pioglitazone selectively activates PPARγ-1. It is about one tenth as potent as rosiglitazone (EC50~500nM for human and mouse PPARγ). In a transactivation assay using COS-1 cells transfected with full length human PPARα and RXRα, pioglitazone and rosiglitazone exhibit low level activation of PPARα at 1µM and 5.4- and 4.2-fold activation, respectively, at a concentration of 10µM.
Product Details
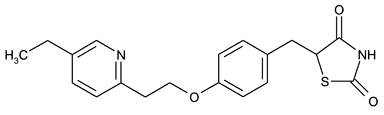
| Formula: | C19H20N2O3S |
| |
| MW: | 356.4 |
| |
| CAS: | 111025-46-8 |
| |
| MI: | 14: 7452 |
| |
| Purity: | ≥97% (HPLC) |
| |
| Identity: | Confirmed by 1H-NMR |
| |
| Appearance: | White to off-white solid. |
| |
| Solubility: | Soluble in DMSO (2.5mg/ml) or dimethyl formamide (2.5mg/ml). |
| |
| Shipping: | Blue Ice |
| |
| Long Term Storage: | -20°C |
| |
| Handling: | Protect from moisture. |
| |
| Scientific Background: | Thiazolidinediones (TZDs) are a group of structurally related PPARγ agonists with anti-diabetic actions in vivo. Rosiglitazone (Prod. No. ALX-350-125) is a prototypical TZD and has served as a reference compound for this class of PPARγ ligands. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Endoplasmic reticulum stress increases LECT2 expression via ATF4: C.Y. Park, et al.; Biochem. Biophys. Res. Commun.
585, 169 (2021),
Abstract;
Cigarette Smoke Extract Modulates Functions of Peroxisome Proliferator-Activated Receptors: M. Matsushita, et al.; Biol. Pharm. Bull.
42, 1628 (2019),
Abstract;
Full Text
Effect of zinc supplements in the attenuated cardioprotective effect of ischemic preconditioning in hyperlipidemic rat heart: S.K. Kansal, et al.; Naunyn Schmiedebergs Arch. Pharmacol.
388, 635 (2016),
Application(s): Cell culture,
Abstract;
Full Text
Rosiglitzone Suppresses Angiotensin II-Induced Production of KLF5 and Cell Proliferation in Rat Vascular Smooth Muscle Cells: D. Gao, et al.; PLoS One
10, e0123724 (2015),
Application(s): Cell Culture,
Abstract;
Full Text
Activation of human peroxisome proliferator-activated receptor (PPAR) subtypes by pioglitazone: J. Sakamoto, et al.; BBRC
278, 704 (2000),
Abstract;
The PPARs: from orphan receptors to drug discovery: T.M. Willson, et al.; J. Med. Chem.
43, 527 (2000),
Abstract;
The structure-activity relationship between peroxisome proliferator-activated receptor gamma agonism and the antihyperglycemic activity of thiazolidinediones: T.M. Willson, et al.; J. Med. Chem.
39, 665 (1996),
Abstract;
An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma): J.M. Lehmann, et al.; J. Biol. Chem.
270, 12953 (1995),
Abstract;
Full Text
[[omega-(Heterocyclylamino)alkoxy]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents: B.C. Cantello, et al.; J. Med. Chem.
37, 3977 (1994),
Abstract;
Related Products