What is protein aggregation and what factors influence protein aggregation?
Protein aggregation is undesired interaction between protein monomers and can be caused by factors such as pH, ionic strength, mechanical agitation, protein concentration, freeze-thaw operations, temperature, and packaging materials and agents (such as glass and silicone oil). Five major mechanisms of aggregation have been identified: concentration, conformational change, chemical reaction, nucleation, and surface-induced aggregation. Surfactants are used in biotechnology to stabilize therapeutic proteins, and inhibit aggregation by preventing protein adsorption on surfaces. Aggregation can be inhibited by kosmotropic salts such as ammonium or magnesium sulfate which stabilize the native protein state.
How does protein aggregation affect bioprocess?
Over the past decade, protein-based therapeutics have emerged as a key driver of growth in the pharmaceutical industry. The efficient large scale production of recombinant proteins requires careful handling of the protein during isolation and purification. Proteins are isolated in an environment devoid of potentially stabilizing factors. As a result, many protein samples show loss of function and reduced stability in standard buffer conditions. Stability is an important issue in protein production, since destabilized proteins are susceptible to chemical and physical alteration leading to loss of activity. Chemical alterations are related to covalent modifications such as oxidation and disulfide bond shuffling. Physical changes include protein unfolding, binding to surfaces, and aggregation. Protein aggregation is favored in the food industry since it imparts desirable texture and flavor. However, for pharmacological applications, protein aggregation represents a major obstacle to the development of protein-based pharmaceuticals. During drug formulation, protein aggregation can impact product quality in terms of biological activity and immunogenicity.
Protein aggregation can occur at all stages in the manufacturing process including cell culture, purification, formulation, storage, shipping, and handling. It can be reversible or irreversible, and aggregates can range in size from microscopic to macroscopic. Therefore, the pharmaceutical industry requires improved methods to detect, monitor, and quantify factors governing aggregation during manufacturing. Accepted industry techniques based on light obscuration and microscopy provide particle size and concentration information, but offer limited ability to differentiate among the different types of contaminants routinely found in therapeutic formulations. The pharmaceutical industry would greatly benefit from improved methods to detect, monitor, and quantify factors governing aggregation during manufacturing.
What are efficient methods to detect protein aggregation?
Understanding the fundamental mechanism of aggregation is valuable for identifying the factors underlying the problem and further aid in developing strategies to prevent aggregation.
Biochemical assays for monitoring protein aggregates often rely on ultracentrifugation, size-exclusion chromatography, gel electrophoresis, dynamic light scattering, or turbidity measurements. These techniques are not applicable for every protein, nor are the assays ideal for addressing a wide range of aggregation problems encountered during formulation development and the manufacturing of protein pharmaceuticals. Currently, there is no accepted method to effectively and simultaneously size, enumerate and classify particles within the critical size range of 1-100 µm. To address this need, scientists at Enzo developed the PROTEOSTAT® reagent, a fluorescent dye that facilitates detection and quantification of aggregated proteins in solution. This dye is an example of a molecular rotor-type fluorochrome which exhibits a significant increase in fluorescence quantum yield in the presence of protein aggregates. As silicone oil micro-droplets are a common contaminant in protein-based therapeutics, we developed a flow cytometry protocol relying on the use of the PROTEOSTAT® detection reagent together with the Bodipy® dye to simultaneously examine protein aggregates and discriminate them from other particles. Fluorescence-based protein stability screens were developed as a suitable alternative as they allow high-throughput screening of many conditions in a 96-well format. This high-throughput screening capability allows for the optimization of different protein drug formulations while ensuring the production of the most stable and particle-free formulations.
Enzo‘s
PROTEOSTAT® Protein Aggregation Assay provides a simple, homogenous assay format for monitoring peptide and protein aggregation. It can be used in a flow cytometry or fluorescence application to determine optimal storage conditions and formulations for proteins, for screening compounds that promote or inhibit protein aggregation, and potentially for measuring molecular chaperone activity in a sensitive manner. The assay can be employed to streamline protein processing and formulation optimization procedures. Relative to conventional protein aggregation detection dyes, such as Thioflavin T, Enzo’s PROTEOSTAT® detection reagent detects visible to subvisible aggregates from a broader range of proteins, yields a much brighter signal, provides at least two orders of magnitude linear dynamic range, and offers superior performance across a broad range of pH values (4-10) and buffer compositions. Sensitivity for this assay is in the sub-micromolar range and as little as 1-5% protein aggregate is detectable in a concentrated protein solution. The assay is capable of providing quantitative analysis of protein aggregation in a robust and high-throughput fashion (Z’ factor score >0.5). Lyophilized native and aggregated protein are provided as negative and positive controls for monitoring changes in protein aggregation status.
PROTEOSTAT® Protein Aggregation Standards kit is the only commercially available protein aggregation standards assay with stabilized, high-quality reference samples for generating trace protein aggregate levels in concentrated monomeric IgG.
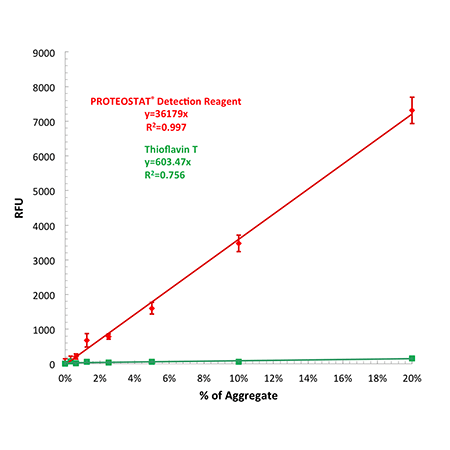 Figure 1. Effective linear dynamic range for antibody aggregate detection using PROTEOSTAT® detection reagent compared with Thioflavin T. Relative fluorescence unit values (RFUs) may differ depending upon the microplate reader employed for the analysis.
Figure 1. Effective linear dynamic range for antibody aggregate detection using PROTEOSTAT® detection reagent compared with Thioflavin T. Relative fluorescence unit values (RFUs) may differ depending upon the microplate reader employed for the analysis.
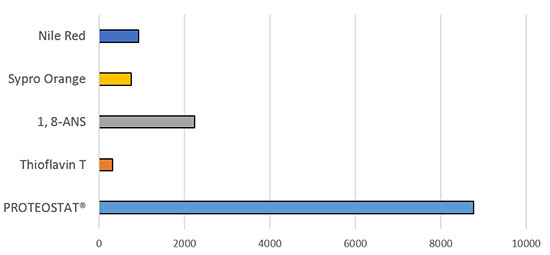 Figure 2. Enzo’s PROTEOSTAT® Protein Aggregation Assay offers superior detection of subvisible protein aggregates compared to conventional dyes.
Figure 2. Enzo’s PROTEOSTAT® Protein Aggregation Assay offers superior detection of subvisible protein aggregates compared to conventional dyes.
Enzo’s catalog of widely cited and thoroughly validated products includes our PROTEOSTAT® assays for contamination monitoring and process optimization. The table below highlights related products available for Protein Aggregation detection. Please check out our
Bioprocess Optimization platform for more information or contact our
Technical Support Team for further assistance.













