Product Details
| Alternative Name: | Superoxide Dismutase (Cu-Zn), Superoxide dismutase 1, SOD1 |
| |
| Host: | Rabbit |
| |
| Immunogen: | Native human Cu/Zn SOD. |
| |
| UniProt ID: | P00441 |
| |
| Source: | Purified from rabbit serum. |
| |
| Species reactivity: | Human, Mouse, Rat
Clam
|
| |
| Applications: | IF, IHC (PS), IP, WB
|
| |
| Recommended Dilutions/Conditions: | Western Blot (1:1,000, colorimetric)
Suggested dilutions/conditions may not be available for all applications.
Optimal conditions must be determined individually for each application. |
| |
| Application Notes: | Detects a band of ~23kDa (human) or ~19kDa (mouse and rat) by Western blot. |
| |
| Purity Detail: | Protein A-affinity purified. |
| |
| Formulation: | Liquid. In PBS containing 50% glycerol and 0.09% sodium azide. |
| |
| Handling: | Avoid freeze/thaw cycles. |
| |
| Shipping: | Blue Ice |
| |
| Long Term Storage: | -20°C |
| |
| Scientific Background: | Superoxide dismutase (SOD) is responsible for the elimination of cytotoxic active oxygen by catalyzing the dismutation of the superoxide radical to oxygen and hydrogen peroxide. There are three SOD isoenzymes in mammalian cells: extracellular SOD (EC SOD, SOD-3); copper and zinc-containing SOD (Cu/Zn SOD, SOD-1); and manganese-containing SOD (Mn SOD, SOD-2). The Cu/Zn form binds Cu and Zn ions and exists as a soluble 32kDa homodimer in the cytosol. Mutations in the SOD-1 gene accounts for approximately 20% of amyelotrophic lateral sclerosis (ALS) or ‘Lou Gehrig disease,’ a neurodegenerative disorder characterized by the death of motor neurons in the brain, brainstem, and spinal cord that results in fatal paralysis. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
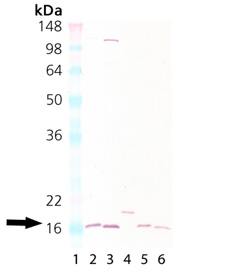
Western Blot Analysis of Cu/Zn SOD: Lane 1:MWM Lane 2: Rat liver tissue lysate, Lane 3: Mouse liver tissue lysate, Lane 4: HeLa, (cell lysate) (Prod No. ADI-LYC-HL100), Lane 5: Brain (mouse), (tissue extract) (Prod No. ADI-LYT-MB100), Lane 6: Brain (rat), (tissue extract) (Prod No. ADI-LYT-RB100).
Please mouse over
General Literature References
Effect of exposure to benzo[a]pyrene on SODs, CYP1A1/1A2- and CYP2E1 immunopositive proteins in the blood clam Scapharca inaequivalvis: M. Monari, et al.; Mar. Environ. Res.
63, 200 (2007),
Abstract;
Mutant Cu/Zn-superoxide dismutase proteins have altered solubility and interact with heat shock/stress proteins in models of amyotrophic lateral sclerosis: G.A. Shinder, et al.; J. Biol. Chem.
276, 12791 (2001),
Abstract;
Amyotrophic lateral sclerosis and structural defects in Cu,Zn superoxide dismutase: H.X. Deng, et al.; Science
261, 1047 (1993),
Abstract;
Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis: D.R. Rosen, et al.; Nat Med.
362, 59 (1993),
Abstract;
---: N. Kurobe, et al.; Biom. Res. 11, 187 (1990),
Sensitive enzyme immunoassay for human Mn superoxide dismutase: N. Kurobe, et al.; Clin. Chim. Acta
192, 171 (1990),
Abstract;
Related Products