Replaces Prod. #: ALX-340-038
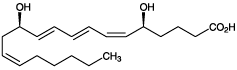
Activator of PPARα. Stimulates leukocyte functions including chemokinesis, chemotaxis, lysosomal enzyme release, adhesion, stimulation of ion fluxes, superoxyde anion production and C3b receptor expression. Induces vascular permeability, NK cell activity, and broncho-constriction. UVmax: 270 nm (50,000).
Product Details
| Alternative Name: | LTB4, 5,12-Dihydroxy-[S-[R*,S*-(E,Z,Z,E)]]-6,8,10,14-eicosatetraenoic acid |
| |
| Formula: | C20H32O4 |
| |
| MW: | 336.5 |
| |
| Source: | Synthetic. |
| |
| CAS: | 71160-24-2 |
| |
| MI: | 14: 5455 |
| |
| Concentration: | 50 µg size: 50 µg/ml
1 mg size: 1 mg/ml |
| |
| Formulation: | Oil dissolved in ethanol. |
| |
| Purity: | ≥97% (HPLC) |
| |
| Appearance: | Colorless liquid. |
| |
| Shipping: | Dry Ice |
| |
| Long Term Storage: | -80°C |
| |
| Use/Stability: | Stable for at least 1 year after receipt when stored, as supplied, at -80°C. |
| |
| Handling: | Open ampule and aliquot into suitable tubes or vials. Store at -80°C or colder. For experiments with cells or tissues, aliquots of ethanol solutions may be added directly to aqueous media. For HPLC, aliquots of ethanol solutions should be evaporated to dryness under a stream of nitrogen and HPLC mobile phase added to the desired concentration. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Ligand selectivity of the peroxisome proliferator-activated receptor alpha: Q. Lin et al.; Biochemistry
38, 185 (1999),
Abstract;
Leukotriene B4: S.W. Crooks & R.A. Stockley; nt. J. Biochem. Cell Biol.
30, 173 (1998),
Abstract;
The peroxisome proliferator-activated receptor alpha (PPARalpha) ligand WY 14,643 does not interfere with leukotriene B4 induced adhesion of neutrophils to endothelial cells: M. Heimburger & J. Palmblad; Biochem. Biophys. Res. Commun.
249, 371 (1998),
Abstract;
The PPARalpha-leukotriene B4 pathway to inflammation control: P.R. Devchand et al.; Nature
384, 39 (1996),
Abstract;
Biosynthesis and biological activity of leukotriene B4: P. Borgeat & P.H. Naccache; Clin. Biochem.
23, 459 (1990),
Abstract;
Leukotriene B4 in inflammation: A.W. Ford-Hutchinson; Crit. Rev. Immunol.
10, 1 (1990),
Abstract;
Leukotrienes and inflammation: S.D. Brain & T.J. Williams; Pharmacol. Ther.
46, 57 (1990),
Abstract;
Quantitative gas chromatography-mass spectrometry analysis of leukotriene B4: W.R. Mathews; Methods Enzymol.
187, 76 (1990),
Abstract;
Leukotrienes and the immune system: M. Rola-Pleszczynski; J. Lipid Mediat.
1, 149 (1989),
Abstract;
Leukotriene B4 and inflammatory disease: R.M. McMillan & S.J. Foster; Agents Actions
24, 114 (1988),
Abstract;
Molecular and cellular properties of human polymorphonuclear leukocyte receptors for leukotriene B4: D.W. Goldman et al.; Fed. Proc.
46, 200 (1987),
Abstract;
Solid-phase extraction and high-performance liquid chromatography analysis of lipoxygenase pathway products: J.D. Eskra et al.; Anal. Biochem.
154, 332 (1986),
Abstract;
The combined use of isolated strips of guinea-pig lung parenchyma and ileum as a sensitive and selective bioassay for leukotriene B4: M.N. Samhoun & P.J. Piper; Prostaglandins
27, 711 (1984),
Abstract;