Replaces Prod. #: ALX-420-003
Stable analog of endogenous S-nitroso compounds. Nitric oxide (NO) donor and a source of NO in vivo which spontaneously releases NO under physiological conditions. The half-life time is 4.6 hours in aqueous media. Potent vasodilator in vitro and in vivo that does not induce pharmacological tolerance. Relaxes isolated bovine coronary artery rings with an EC50=0.1µM. It also inhibits vascular smooth muscle cell mitogenesis and proliferation, inhibits leukocyte adherence to endothelium, and inhibits cysteine proteases. The unique NO releasing properties of SNAP make it an ideal agent for studying the pharmacological and physiological actions of NO.
Product Details
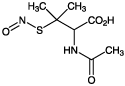
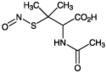
| Alternative Name: | S-Nitroso-N-acetyl-D,L-penicillamine |
| |
| Formula: | C7H12N2O4S |
| |
| MW: | 220.3 |
| |
| Source: | Synthetic. |
| |
| CAS: | 79032-48-7 |
| |
| Purity: | ≥98% (HPLC) |
| |
| Appearance: | Green crystalline solid. |
| |
| Solubility: | Soluble in DMSO (25mg/ml) 100% ethanol (25mg/ml) or methanol; slightly soluble in water (2mg/ml). |
| |
| Shipping: | Ambient Temperature |
| |
| Long Term Storage: | -20°C |
| |
| Use/Stability: | Stable for up to 1 year after receipt when stored at -20°C. |
| |
| Technical Info/Product Notes: | Note: Product is not sterile. |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
Please mouse over
Product Literature References
Dysregulated Expression of Arginine Metabolic Enzymes in Human Intestinal Tissues of Necrotizing Enterocolitis and Response of CaCO2 Cells to Bacterial Components: K.T. Leung, et al.; J. Nutr. Biochem.
29, 64 (2016),
Application(s): NO supplementation in CaCO2 cells,
Abstract;
Molecular Magnetic Resonance Imaging of Angiogenesis In Vivo using Polyvalent Cyclic RGD-Iron Oxide Microparticle Conjugates : S. Melemenidis, et al.; Theranostics
5, 515 (2015),
Application(s): Cell Culture,
Abstract;
Full Text
SNAP, a NO donor, induces cellular protection only when cortical neurons are submitted to some aggression process: S. Figueroa, et al.; Brain Res.
1034, 25 (2005),
Abstract;
SNAP, a NO donor, induces cortical neuron death by a mechanism in which the caspase pathway is implicated: S. Figueroa, et al.; Brain Res.
1047, 168 (2005),
Abstract;
The reaction of S-nitroso-N-acetyl-D,L-penicillamine (SNAP) with the angiotensin converting enzyme inhibitor, captopril--mechanism of transnitrosation: D.V. Aquart & T.P. Dasgupta; Org. Biomol. Chem.
3, 1640 (2005),
Abstract;
In vitro cytotoxicity of glyco-S-nitrosothiols. a novel class of nitric oxide donors: H. Babich & H.L. Zuckerbraun; Toxicol. In Vitro
15, 181 (2001),
Abstract;
Nitric oxide- and nitric oxide donors-induced relaxation and its modulation by oxidative stress in piglet pulmonary arteries: J.G. Lopez-Lopez, et al.; Br. J. Pharmacol.
133, 615 (2001),
Abstract;
Full Text
In vitro cytotoxicity of the nitric oxide donor, S-nitroso-N-acetyl- penicillamine, towards cells from human oral tissue: H. Babich, et al.; Pharmacol. Toxicol.
84, 218 (1999),
Abstract;
Modulation of S-nitroso-N-acetyl-D,L-penicillamine (SNAP) induced HL-60 cell death by tetrahydrobiopterin: S. Kojima, et al.; Anticancer Res.
17, 929 (1997),
Abstract;
Nitric oxide donor SNAP induces apoptosis in smooth muscle cells through cGMP-independent mechanism: E. Nishio, et al.; BBRC
221, 163 (1996),
Abstract;
An investigation of some S-nitrosothiols, and of hydroxy-arginine, on the mouse anococcygeus: A. Gibson, et al.; Br. J. Pharmacol.
107, 715 (1992),
Abstract;
Lack of tolerance to a 24-hour infusion of S-nitroso N- acetylpenicillamine (SNAP) in conscious rabbits: J.E. Shaffer, et al.; J. Pharmacol. Exp. Ther.
260, 286 (1992),
Abstract;
Production of hydroxyl radicals from the simultaneous generation of superoxide and nitric oxide: N. Hogg, et al.; Biochem. J.
281, 419 (1992),
Abstract;
S-nitroso-glutathione inhibits platelet activation in vitro and in vivo: M.W. Radomski, et al.; Br. J. Pharmacol.
107, 745 (1992),
Abstract;
Differential hemodynamic effects and tolerance properties of nitroglycerin and an S-nitrosothiol in experimental heart failure: J.A. Bauer & H.-L. Fung; J. Pharmacol. Exp. Ther.
256, 249 (1991),
Abstract;
Mechanism of vascular smooth muscle relaxation by organic nitrates, nitrites, nitroprusside and nitric oxide: evidence for the involvement of S-nitrosothiols as active intermediates: L.J. Ignarro, et al.; J. Pharmacol. Exp. Ther.
218, 739 (1981),
Abstract;