Replaces Prod. #: BML-EI175
Phospholipase A2 (PLA2) inhibitor active against the enzymes found in many snake venoms as well as those of human platelets and synovial fluids. Inhibits ionophore-stimulated PLA2 activity in human neutrophils. Has been shown to be nephropathic and carcinogenic.
Product Details
| Alternative Name: | 8-Methoxy-6-nitrophenanthrol(3,4-d)-1,3-dioxide-5-carboxylic acid, 6-Nitrophenanthrol(3,4-d)-1,3-dioxide-5-carboxylic acid |
| |
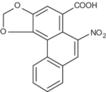
| Formula: | C17H11NO7 (Aristolochic acid I) and C16H9NO6 (Aristolochic acid II) |
| |
| MW: | 341.3 (Aristolochic acid I) and 311.3 (Aristolochic acid II) |
| |
| Source: | Isolated from Aristolochia. |
| |
| CAS: | 313-67-7 (Aristolochic acid I), 475-80-9 (Aristolochic acid II), 67123-64-2 (mixture) |
| |
| MI: | 14: 786 |
| |
| RTECS: | CF3325000 |
| |
| Purity: | ≥97% (~1:1 mixture of aristolochic acids I (A) and II (B)) |
| |
| Appearance: | Yellow solid. |
| |
| Solubility: | Soluble in DMSO or 100% ethanol. Sparingly soluble in water. |
| |
| Shipping: | Ambient Temperature |
| |
| Long Term Storage: | Ambient |
| |
| Regulatory Status: | RUO - Research Use Only |
| |
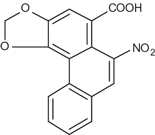
Aristolochic acid II
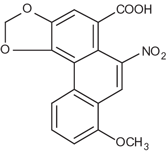
Aristolochic acid I
Please mouse over
Product Literature References
Misexpression of FATTY ACID ELONGATION1 in the Arabidopsis epidermis induces cell death and suggests a critical role for phospholipase A2 in this process: J.J. Reina-Pinto, et al.; Plant Cell
21, 1252 (2009),
Abstract;
Full Text
Effect of aristolochic acid on intracellular calcium concentration and its links with apoptosis in renal tubular cells: Y.H. Hsin, et al.; Apoptosis
11, 2167 (2006),
Abstract;
Aristolochic acid and ’Chinese herbs nephropathy’: a review of the evidence to date: J.P. Cosyns; Drug Saf.
26, 33 (2003), Review,
Abstract;
Detection of DNA adducts formed by aristolochic acid in renal tissue from patients with Chinese herbs nephropathy: H.H. Schmeiser, et al.; Cancer Res.
56, 2025 (1996),
Abstract;
Selective inhibition of group II phospholipase A2 by quercetin: M. Lindahl & C. Tagesson; Inflammation
17, 573 (1993),
Abstract;
Evidence for different mechanisms involved in the formation of lyso platelet-activating factor and the calcium-dependent release of arachidonic acid from human neutrophils: J.D. Winkler, et al.; Biochem. Pharmacol.
44, 2055 (1992),
Abstract;
The effects of the phospholipase A2 inhibitors aristolochic acid and PGBx on A23187-stimulated mobilization of arachidonate in human neutrophils are overcome by diacylglycerol or phorbol ester: M.D. Rosenthal, et al.; Biochim. Biophys. Acta
1126, 319 (1992),
Abstract;
Suramin alters phosphoinositide synthesis and inhibits growth factor receptor binding in HT-29 cells: R. Kopp & A. Pfeiffer; Cancer Res.
50, 6490 (1990),
Abstract;
Effects of aristolochic acid on phospholipase A2 activity and arachidonate metabolism of human neutrophils: M.D. Rosenthal, et al.; Biochim. Biophys. Acta
1001, 3 (1989),
Abstract;
Edema-inducing activity of phospholipase A2 purified from human synovial fluid and inhibition by aristolochic acid: B.S. Vishwanath, et al.; Inflammation
12, 549 (1988),
Abstract;
Characterization of three edema-inducing phospholipase A2 enzymes from habu (Trimeresurus flavoviridis) venom and their interaction with the alkaloid aristolochic acid: B.S. Vishwanath, et al.; Toxicon
25, 501 (1987),
Abstract;
Interaction of aristolochic acid with Vipera russelli phospholipase A2: its effect on enzymatic and pathological activities: B.S. Vishwanath & T.V. Gowda; Toxicon
25, 929 (1987),
Abstract;
Interaction of phospholipase A2 from Vipera russelli venom with aristolochic acid: a circular dichroism study: B.S. Vishwanath, et al.; Toxicon
25, 939 (1987),
Abstract;