Replaces Prod. #: ALX-201-273
Product Details
| Alternative Name: | HspB1, Heat shock protein 27 |
| |
| Recommended Dilutions/Conditions: | Western Blot (100ng, colorimetric)
Suggested dilutions/conditions may not be available for all applications.
Optimal conditions must be determined individually for each application. |
| |
| MW: | ~27kDa |
| |
| Source: | Produced in E. coli. |
| |
| UniProt ID: | P04792 |
| |
| GenBank ID: | L39370 |
| |
| Formulation: | Liquid. In 20mM TRIS, pH 7.5, containing 10mM sodium chloride, 1mM EDTA, and 1mM DTT. |
| |
| Purity: | ≥90% (SDS-PAGE; Western blot) |
| |
| Purity Detail: | Purified by multi-step chromatography. |
| |
| Applications: | WB
|
| |
| Application Notes: | Western blot control. |
| |
| Shipping: | Dry Ice |
| |
| Long Term Storage: | -80°C |
| |
| Scientific Background: | Human Hsp27, mouse Hsp25 and ab-crystallin are part of a diverse family of small heat shock proteins which are produced in all organisms. They function as chaperone-like proteins by binding unfolded polypeptides and preventing uncontrolled protein aggregation. Hsp27 is believed to exist mainly as oligomers of as many as 8-40 Hsp27 protein monomers in cells. Data suggests that the large oligomers of Hsp27 have a chaperone-like activity by serving as a site where unfolded proteins may bind until ATP and Hsp70-dependent refolding can occur. The phosphorylation and oligomerization state of Hsp27 has been suggested to regulate microfilament organization, since only the nonphosphorylated lower molecular weight forms of Hsp27 bind actin barbed ends and inhibit polymerization. Hsp27 is also believed to protect cells by enhancing cellular glutathione levels, as elevated glutathione levels have been measured in cells overexpressing Hsp27. Data from studies using wild-type Hsp27 and mutant forms in which the serine phosphorylation sites were |
| |
| Regulatory Status: | RUO - Research Use Only |
| |

Western Blot analysis: Lane 1: MWM, Lane 2: HSP27 probed with HSP27 mAb.

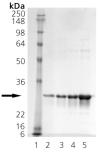
SDS-PAGE analysis: Lane 1: MW marker, Lane 2: 0.5µg, Lane 3: 1µg, Lane 4: 2µg, Lane 5: 5µg HSP27 stained with Imperial stain.
Please mouse over
Product Literature References
The cellular stress response of rat skeletal muscle following lengthening contractions: E. Pollock-Tahiri, et al.; Appl. Physiol. Nutr. Metab.
42, 708 (2017),
Abstract;
Retinoic acid induces nuclear FAK translocation and reduces breast cancer cell adhesion through Moesin, FAK, and Paxillin: A. M. Sanchez, et al.; Mol. Cell. Endocrinol.
430, 1 (2016),
Application(s): Immunoblotting,
Abstract;
Hsp25 and Hsp72 content in rat skeletal muscle following controlled shortening and lengthening contractions: A.M. Holwerda, et al.; Appl. Physiol. Nutr. Metab.
39, 1380 (2014),
Abstract;
The effect of heat stress on skeletal muscle contractile properties: M. Locke, et al.; Cell Stress Chaperones
19, 519 (2014),
Abstract;
Full Text
Tumor imaging and targeting potential of an Hsp70-derived 14-mer peptide: M. Gehrmann, et al.; PLoS One
9, e105344 (2014),
Abstract;
Full Text
Antigen microarrays identify unique serum autoantibody signatures inclinical and pathologic subtypes of multiple sclerosis: H. Weiner, et al. ; PNAS
105, 18889 (2008),
Application(s): Antigen Microarray ,
Abstract;
Maximal eccentric exercise induces a rapid accumulation of small heat shock proteins on myofibrils and a delayed HSP70 response in humans: G. Paulsen, et al.; Am. J. Physiol. Regul. Integr. Comp. Physiol.
293, R844 (2007),
Application(s): EIA using human vastus lateralis muscle tissue,
Abstract;
Full Text
Methylglyoxal modifies heat shock protein 27 in glomerular mesangial cells: R.H. Nagaraj, et al. ; FEBS Lett.
551, 113 (2003),
Application(s): In Vitro Assay ,
Abstract;
Evidence for a role of p38 MAP kinase in expression of alkaline phosphatase during osteoblastic cell differentiation: J. Caverzasio, et al. ; Bone
30, 91 (2002),
Application(s): In Vitro Assay ,
Abstract;
Multiple mitogenic pathways in pancreatic cancer cells are blocked by a truncated epidermal growth factor receptor: M. Korc, et al. ; Cancer Res.
62, 5611 (2002),
Abstract;
Activation of p38MAPK by TGF-beta in fetal rat hepatocytes requires radical oxygen production, but is dispensable for cell death: I. Fabregat, et al. ; FEBS Lett.
499, 225 (2001),
Application(s): In Vitro Assay ,
Abstract;
Follicle-stimulating hormone stimulates protein kinase A-mediated histone H3 phosphorylation and acetylation leading to select gene activation in ovarian granulosa cells: M. Hunzicker-Dunn, et al. ; J. Biol. Chem.
276, 40146 (2001),
Application(s): WB ,
Abstract;
TNF-alpha inhibits UCP-1 expression in brown adipocytes via ERKs. Opposite effect of p38MAPK: A. Porras, et al. ; FEBS Lett.
493, 11 (2001),
Application(s): In Vitro Assay ,
Abstract;
Related Products